|
5/29/2023 0 Comments Amorphous vs crystalline
However, atoms can have a short-range order due to chemical bonding characteristics.įigure 01: A Diagram showing Crystalline and Amorphous Solid Structures Upon cooling, the atoms arrange in a disordered manner to produce glass thus, we call it as amorphous. At high temperatures, these materials melt together, and when we cool them, rigid glass forms rapidly. We make glass primarily with sand (silica/ SiO 2), and bases like sodium carbonate, and calcium carbonate. Moreover, glass, gels, thin films, plastics and nanomaterials are some examples of this type of solids. There, it does not have a long-range ordered arrangement of atoms, molecules, or ions within its structure. Side by Side Comparison – Amorphous vs Crystalline Solid in Tabular FormĪmorphous solid is a form of solid which lacks a crystalline structure. 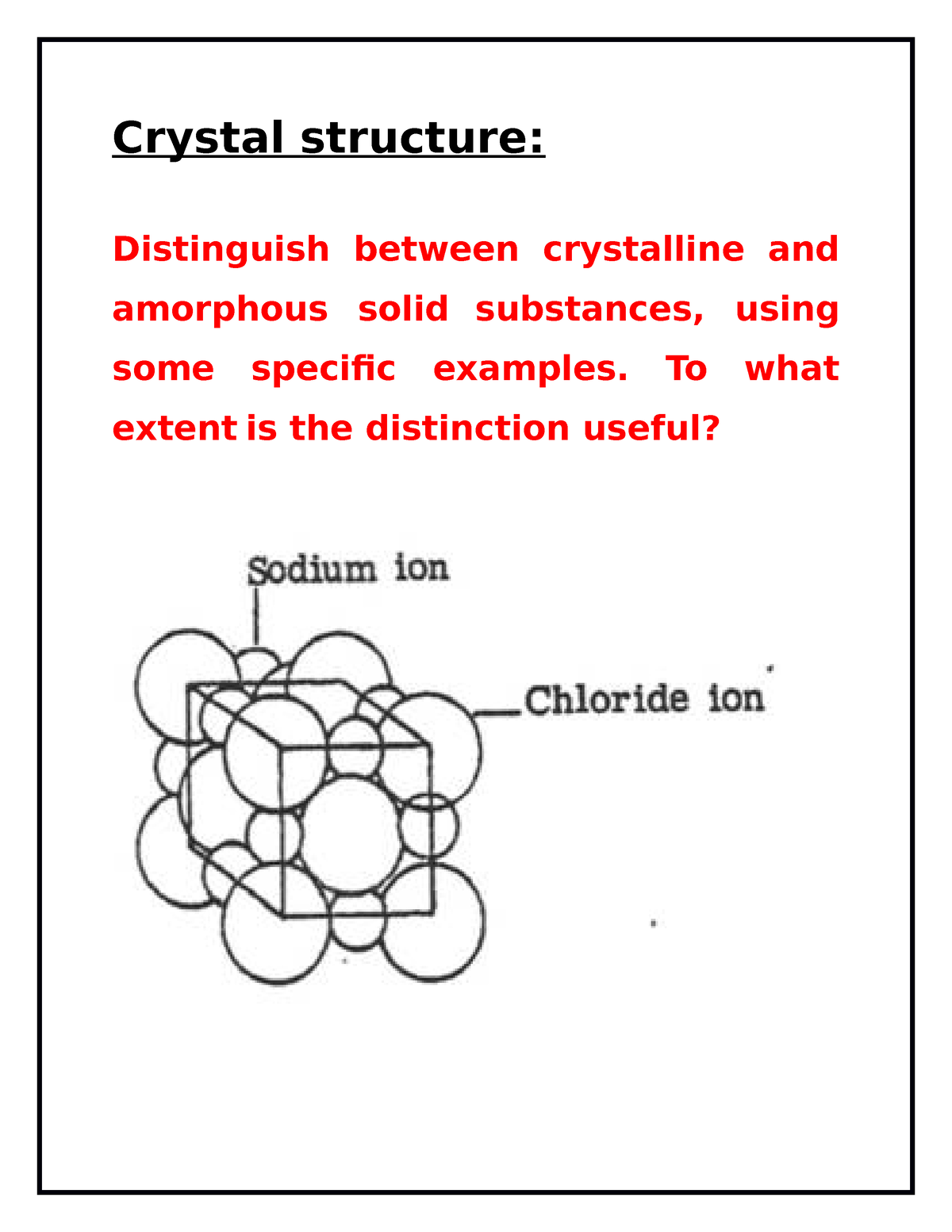
Depending on the need, we can prepare both types separately. 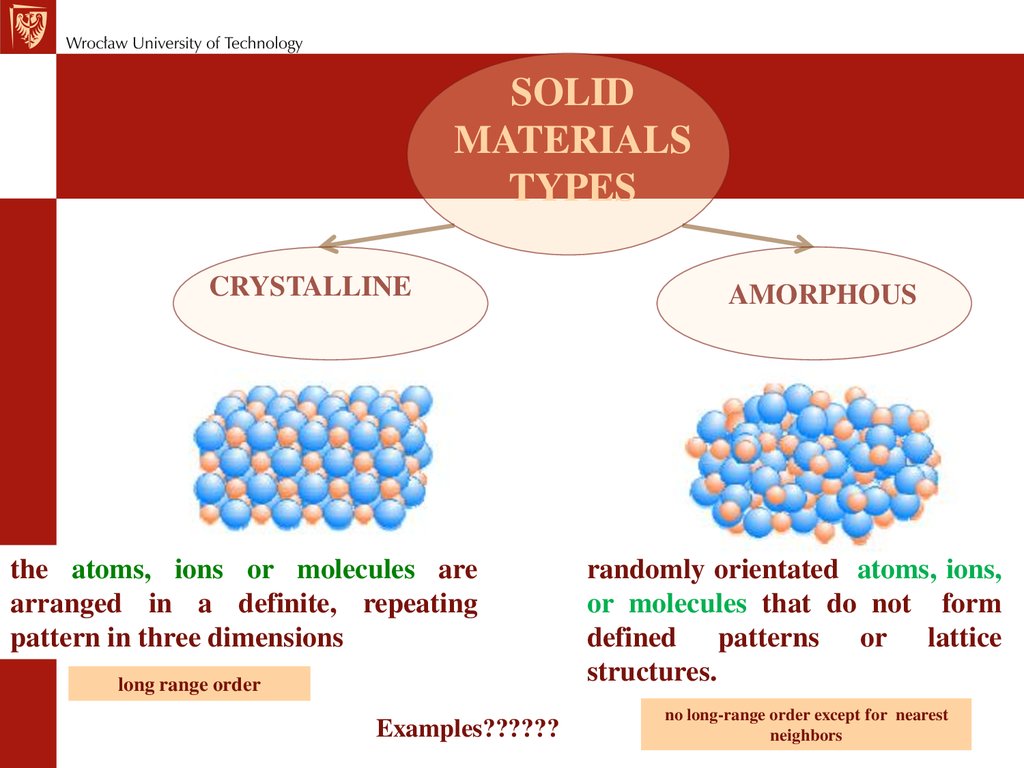
However, some solids are present in both crystalline and amorphous forms. We can classify solids into two as crystalline and amorphous depending on the atomic level arrangement. The key difference between amorphous and crystalline solid is that the crystalline solids have an ordered long-range arrangement of atoms or molecules within the structure, whereas the amorphous solids lack ordered long-range arrangement.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
June 2023
Categories |
 RSS Feed
RSS Feed
